Independent CRO
Clinical trials where
imaging defines
the outcome.
Berlin · Philadelphia · Est. 2006
Imaging core lab, full CRO management, and consulting — across therapeutic areas and all development phases.

Independent CRO
Berlin · Philadelphia · Est. 2006
Imaging core lab, full CRO management, and consulting — across therapeutic areas and all development phases.
For twenty years, we have been the team sponsors call when imaging is not a secondary endpoint — but the reason the study exists.

Imaging protocol, site qualification, DICOM logistics, and independent reads are managed by the same team running your trial — not contracted out to a separate imaging vendor. Most sponsors coordinate a CRO and an imaging core lab independently. We are both — one team, one point of accountability, from protocol to final read.
From imaging charter and site qualification through DICOM image collection, quality control, and blinded reads to the final results dataset — we manage every imaging step with the rigour that regulatory submissions demand. Response criteria covered include RECIST 1.1, iRECIST, PERCIST, and study-specific frameworks.
Two decades of direct experience in imaging-based trials across therapeutic areas and development phases. We help sponsors design programs that satisfy regulatory expectations and prepare for productive interactions with FDA and EMA.
When a programme requires expertise beyond imaging — biostatistics, regulatory writing, toxicology, CMC — pharmtrace works through CONELIS, a Berlin-based network of independent experts in pharmaceutical development. Members can be engaged individually or assembled as interdisciplinary teams, on demand, without the overhead of a large organisation. conelis.org ↗
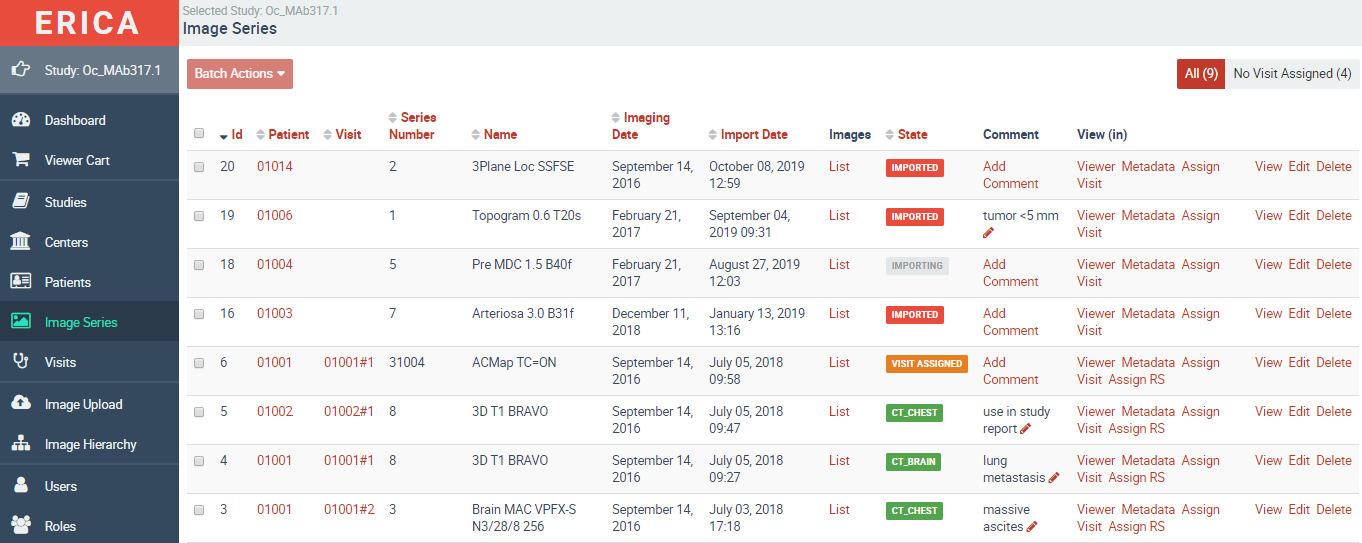
ERICA is our proprietary, fully validated imaging platform — purpose-built for the demands of clinical trials. It covers DICOM image storage, technical and medical quality control, and independent blinded reads in a single, regulatory-compliant environment.
Every project-specific configuration is separately validated. No general-purpose software adapted for clinical use — a system designed from the ground up for this work.

Twenty years. Independent. Private.
Committed to the work.
Twenty years of imaging trials teaches you exactly where they break — and how to prevent it.
Berlin · Philadelphia · Est. 2006
pharmtrace manages clinical trials globally from two offices that work as one team — with direct access to local expertise where it matters, and clear accountability at every step.
Philadelphia, PA
One Liberty Place